Availability of leukopak collection services is subject to certain regions. For more information, please get in touch with us.
Leukopaks
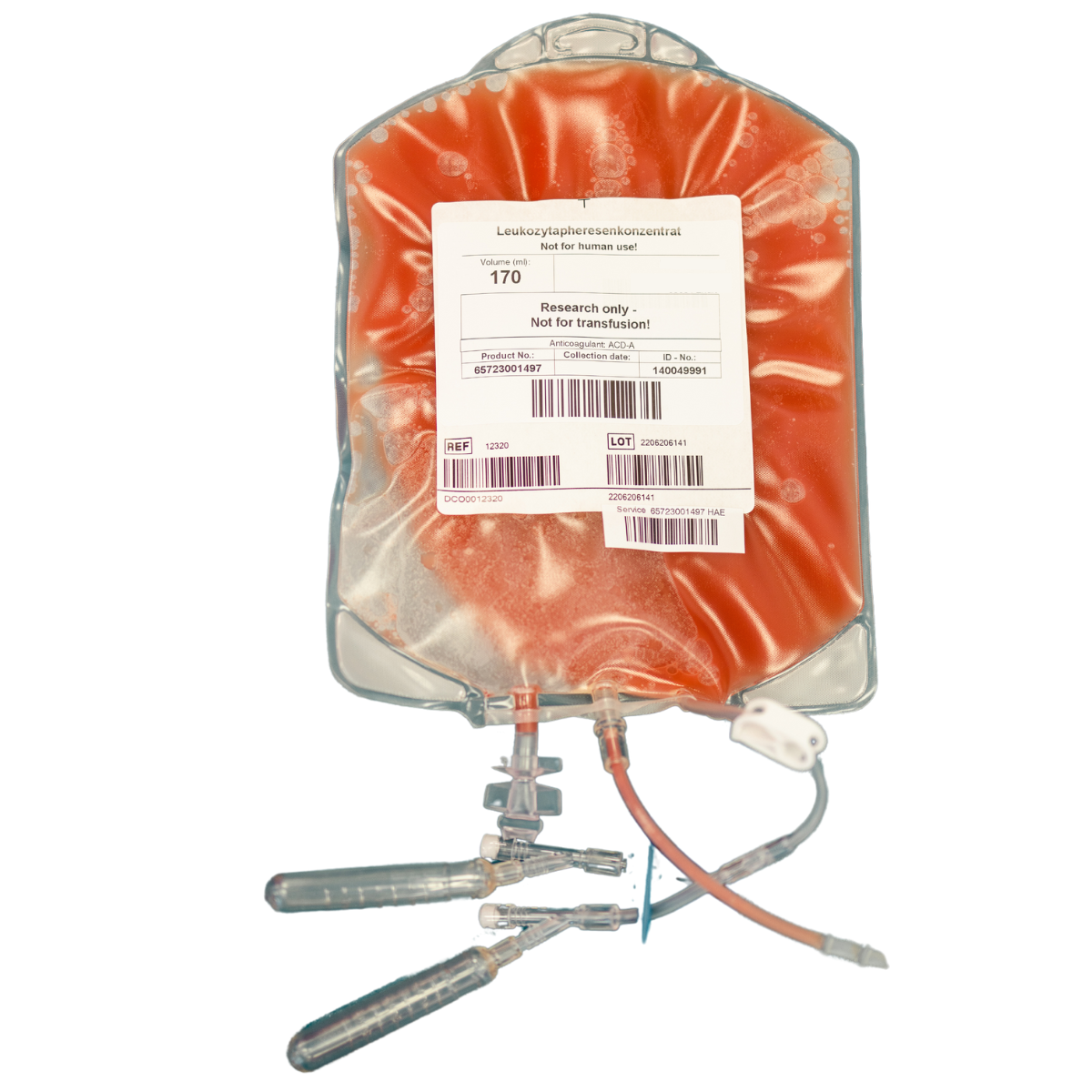
Leukopak is a blood-derived product obtained through apheresis, rich in leukocytes and is used by researchers and manufacturers as starting material in cell therapy.


Leukopak Donor Collection Program
From our robust donor network in Germany and in the USA, donors fulfilling the requirements for leukopak donations are approached to be enrolled within the leukopak donation program.
All donations and manufacturing of the product in Europe is done according to the German guidelines for Collection and Processing of Blood and Blood Components (“Hämotherapierichtlinie”) as well as the PPTA Qualified Donor standard and internal SOPs. A manufacturing license as well as a GMP certificate is granted to all donor centers in Germany.
All donations and manufacturing of the product in the USA is done according to the FDA guidelines for Collection and Processing of Blood and Blood components and internal SOPs. All our donor centers are regularly inspected by authorities and customers.
Fresh Leukopaks
Grifols Bio Supplies offers GMP-grade and research use only (RUO) fresh leukopaks, collected in Europe and the United States. Grifols extensive donor network and expertise in apheresis ensure a consistent supply of starting materials for large-scale manufacturing processes of CAR-T therapies.
Fresh leukopaks are collected from healthy donors without any further manufacturing steps. They are shipped within 24 hours and require immediate use.
Find the full specifications of fresh leukopaks below.
Europe (Germany) or US
Donors have an approved consent for donating blood. All donors are screened to ensure that each of them is in good health condition and that products are not collected from a high-risk population for blood-borne infections
Each donation is tested for the following viral markers: Serologically tested: non-reactive for HBsAg, anti-HBc, HIV-Ag/Ab, HCV-Ab and syphilis. NAT tested: non-reactive for HIV-1; HCV; HBV; HAV; PVB19, HEV.
Each donor is tested for standard FDA viral markers and found negative for anti-HIV Ag/Ab, HBsAg, Anti-HCV, HTLV I/II, Treponema pallidum antibodies, anti-HBc, atypical antibody, HIV-1 RNA/HBV DNA/HCV RNA, WNV-NAT, and Chagas (performed on the donor's first donation)
Age, gender, race, blood group and other
>170 mL
ACD-A
Leukapheresis
≥ 40 x 10⁹ cells/L
≤ 0.5 mmol/L
≥ 5 x 10⁹ cells/L
≥ 28 x 10⁹ cells/L
- R&D cell therapy
- Source of PBMCs
RUO (research use only)
| Fresh leukopak specifications | |
|---|---|
|
Origin of leukopaks |
Europe (Germany) or US |
|
Donor criteria |
Donors have an approved consent for donating blood. All donors are screened to ensure that each of them is in good health condition and that products are not collected from a high-risk population for blood-borne infections |
|
Viral testing for European donors |
Each donation is tested for the following viral markers: Serologically tested: non-reactive for HBsAg, anti-HBc, HIV-Ag/Ab, HCV-Ab and syphilis. NAT tested: non-reactive for HIV-1; HCV; HBV; HAV; PVB19, HEV. |
|
Viral testing for US donors |
Each donor is tested for standard FDA viral markers and found negative for anti-HIV Ag/Ab, HBsAg, Anti-HCV, HTLV I/II, Treponema pallidum antibodies, anti-HBc, atypical antibody, HIV-1 RNA/HBV DNA/HCV RNA, WNV-NAT, and Chagas (performed on the donor's first donation) |
|
Donor data provided |
Age, gender, race, blood group and other |
|
Format |
>170 mL |
|
Anticoagulant |
ACD-A |
|
Obtention method |
Leukapheresis |
|
Leukocyte concentration |
≥ 40 x 10⁹ cells/L |
|
Hemoglobin concentration |
≤ 0.5 mmol/L |
|
Monocyte concentration |
≥ 5 x 10⁹ cells/L |
|
Lymphocyte concentration |
≥ 28 x 10⁹ cells/L |
|
Use |
|
|
Grade |
RUO (research use only) |

Cryopreserved Leukopaks
Cryopreserved leukopaks provided by Grifols have been developed using a consistent cryopreservation process that achieves high cell recovery.
Our cryopreserved leukopaks are produced using our fresh leukopaks collected in Europe, which are split into up to 6 different cryobags and a cryoprotectant medium is added to preserve the cells. The cryopreservation process is performed using a controlled-rate freezer and the material is stored in a nitrogen tank.
The cryopreservation is performed within 24 hours after collection to ensure the viability and recovery of the cells.
Find the full specifications of cryopreserved leukopaks below.
Europe (Germany)
Donors are eligible and deferred according to German and European guidelines and regulations.
Age, gender, race, blood group and other.
Each donation is tested for the following viral markers: serology testing: non-reactive for HBsAg, anti-HBc, HIV-1/2-Ab, HCV-Ab and syphilis. NAT testing: non-reactive for HIV-1, HCV, HBV; HAV; PVB19, HEV, WNV (tested from June to November).
50 mL (±5 mL)
CryoMACS 250 (Miltenyi)
CryoStor® CS10
50 × 106 cells/mL (±10%)
2.5 × 109 (±10%)
- R&D cell therapy
- Source of PBMCs
RUO (research use only)
| Cryopreserved leukopak specifications | |
|---|---|
|
Origin of leukopaks |
Europe (Germany) |
|
Donor criteria |
Donors are eligible and deferred according to German and European guidelines and regulations. |
|
Donor data provided |
Age, gender, race, blood group and other. |
|
Viral testing |
Each donation is tested for the following viral markers: serology testing: non-reactive for HBsAg, anti-HBc, HIV-1/2-Ab, HCV-Ab and syphilis. NAT testing: non-reactive for HIV-1, HCV, HBV; HAV; PVB19, HEV, WNV (tested from June to November). |
|
Package volume |
50 mL (±5 mL) |
|
Cryobag type |
CryoMACS 250 (Miltenyi) |
|
Cryopreservation media |
CryoStor® CS10 |
|
Targeted cell concentration |
50 × 106 cells/mL (±10%) |
|
Targeted cells/cryobag |
2.5 × 109 (±10%) |
|
Viability after thawing |
≥90% |
|
Recovery after thawing |
≥80% |
|
Use |
|
|
Grade |
RUO (research use only) |
Leukopak collection services
Available Leukopaks
Grifols Bio Supplies offers a variety of human leukopaks collected from both Europe (Germany) and the USA, ensuring a broad and reliable supply chain:
- Fresh leukopaks: These are collected from donors in both Europe and the USA without any further manufacturing steps. Fresh leukopaks are shipped within 24 hours and need to be used immediately. We offer both GMP and RUO grade leukopaks.
- Cryopreserved leukopaks: These are collected from donors in Europe and cryopreserved approximately 24 hours post-collection. Cryopreserved leukopaks can be stored for extended periods in the gas phase of liquid nitrogen and can be used as needed. We offer RUO cryo leukopaks.
Let Grifols be your trusted partner for leukopaks
Contact a Bio Supplies representative today to get more information
More Cell Therapy Products
The information contained in these webpages are intended for a professional audience of pharmaceutical and in vitro diagnostic manufacturing personnel. All products are intended for research and development and manufacturing usage.
Availability of the products are subjected to certain regions. For more information, please contact us